Primary surface measurement reported
Sessile-drop contact angle (hydrophobicity) measurements on LIG and nPt-LIG electrode surfaces.
Client Citation Analysis
Sessile-drop contact angle (hydrophobicity) measurements on LIG and nPt-LIG electrode surfaces.
Hydrophobicity was analyzed using a Droplet lab DROPOMETER-M with contact-angle calculations based on the polynomial method (non-axisymmetric drop) from static images captured in sessile droplet mode.
Contact-angle outputs were used to quantify batch-to-batch variation in LIG hydrophobicity immediately after graphitization, compare wettability across multiple testing solutions (including common biological buffers), and evaluate wettability changes after nanoplatinum metallization.
Four unique batches (nine electrodes each) were prepared on individual days by the same operator for batch-to-batch hydrophobicity testing; Figure 1 reports n = 24 for each violin-plot group and n = 6 electrodes in each batch for batch-average plots.
Hydrophobicity reported as contact angle (degrees) measured by goniometry using sessile droplets on LIG and nanoplatinum-metallized LIG (nPt-LIG) electrodes.
The study also reports LIG characterization using stereomicroscopy, open circuit potential (OCP), and cyclic voltammetry (CV) as part of the batch-variation assessment and electrochemical sensing context, including calculation of oxidation peak current and area between anodic/cathodic curves from CV traces.
Hydrophobicity (contact angle) was measured using a Droplet lab DROPOMETER-M by placing a 2 µL aliquot on the working area of the LIG electrode, capturing a static image in sessile droplet mode, and calculating contact angle using a polynomial (non-axisymmetric drop) method. In the software workflow, key image features (e.g., droplet edges/profile of interest) were identified following manufacturer recommendations, and images from each test were archived.
In this study’s batch-variation workflow, the resulting contact-angle data were used to compare hydrophobicity across fabrication batches, across testing solutions (including common buffers), and before/after nanoplatinum metallization.
The average contact angle for LIG in DI was reported as 58.6 ± 1.4°, and the authors interpret this as indicating a hydrophilic surface under their test conditions.
The mean contact angle in 2× isotonic buffer was reported as 59.3 ± 2.6°, and described as not significantly different than DI; within fabrication batches, contact angle variation for non-modified LIG in DI and isotonic bicarbonate was reported as less than 5%.
Contact angles for non-modified LIG in HEPES, MES, and Tris were reported as significantly lower than DI, and the authors interpret this as each buffer behaving as a mild surfactant impacting surface tension; within-group variation for these buffer conditions was reported as 6% to 8%.
When 36 electrodes were fabricated in a single day, reported contact-angle variation was more than 30%. Fabricating and analyzing 36 electrodes as four batches of nine with a 30 min laser downtime between batches was associated with reduced variation (reported as a reduction from 30% to less than 5%), and the authors identify laser maintenance, operational frequency, and batch size as control factors for future protocols.
After metallization with nPt, the contact angle in DI and isotonic carbonate buffer was reported as 78 ± 4°, described as an increase by nearly 20% with a more hydrophobic surface. The authors report the HEPES/MES/Tris contact angles for nPt-LIG as similar (63 ± 2°) and higher than all LIG experiments, with within-batch variation described as 5%.

Shows representative goniometry images for four electrode batches (LIG sample with a 2 µL DI droplet) with calculated contact angles overlaid.
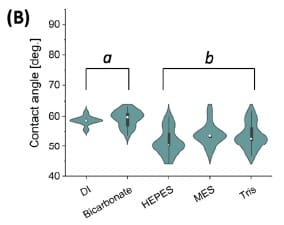
Violin plots compare contact-angle distributions across testing liquids for non-modified LIG (n = 24 per group), with group subsetting indicated by LSD lettering.
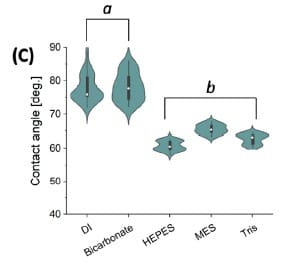
Violin plots show how contact-angle distributions shift after nanoplatinum metallization across the same set of testing liquids (n = 24 per group).
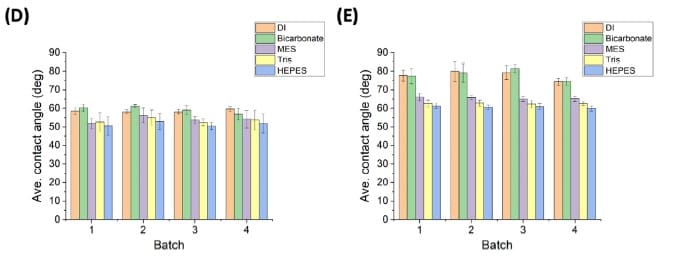
Batch-average contact angles (with standard deviation error bars) are shown for non-modified LIG (D) and nPt-LIG (E), with n = 6 electrodes per batch.
The paper frames laser-inscribed graphene as an emerging platform for electrochemical sensing and positions batch-to-batch variability as a practical challenge for scalable device manufacturing. Within that context, the Droplet lab DROPOMETER-M contact-angle measurements provide a direct readout of LIG hydrophobicity and how it varies with fabrication batching practices.
The authors also use contact-angle comparisons across testing buffers (including zwitterionic buffer components) and after nanoplatinum metallization to discuss interfacial behavior in different solutions, linking wettability differences to how electrodes may behave in common electrochemical sensing environments.
The authors report that preliminary Young–Laplace (axisymmetric) fitting showed significant errors (up to 60% for the same sample tested sequentially), and therefore used the polynomial (non-axisymmetric drop) approach throughout for contact-angle calculations.
The study reports markedly different contact-angle variation for 36 electrodes fabricated in a single day versus splitting fabrication into multiple batches with laser downtime, and highlights maintenance and operational factors (e.g., lens cleaning, operational frequency, batch size) as protocol controls.
HEPES, MES, and Tris produced significantly lower contact angles on non-modified LIG than DI, and the authors interpret these shifts in the context of surfactant-like behavior and interfacial effects.
Nanoplatinum metallization increased reported contact angles (78 ± 4° in DI and isotonic carbonate buffer) and shifted contact angles in HEPES/MES/Tris relative to non-modified LIG.
The authors characterize LIG using goniometry together with electrochemical tests (OCP and CV) as part of the overall analysis of batch-to-batch variation for electrochemical sensing applications.