Primary surface measurement reported
Water contact angle (wettability) of electrospun polystyrene fiber mats (and a PS film reference surface) using water droplets.
Client Citation Analysis
Water contact angle (wettability) of electrospun polystyrene fiber mats (and a PS film reference surface) using water droplets.
The authors state: “The contact angle was analyzed on a tabletop goniometer from Droplet Lab.”
Water contact angle values and images are used to compare hydrophobicity across electrospinning conditions (chloroform:DMF ratio and applied voltage) and to support a Cassie–Baxter treatment of rough-surface wettability reported as fractional projected area (f).
8.4
0.833
1.078
Water contact angle of electrospun PS fiber mats with water, presented as contact-angle images and contact angle versus applied voltage for multiple chloroform:DMF solvent ratios.
Fiber morphology (including fiber and pore dimensions) by SEM, plus solution electrical conductivity and viscosity; surface area and porosity were obtained via gas adsorption and discussed alongside wettability results.
The paper uses the Droplet Lab tabletop goniometer to analyze water contact angle on electrospun PS fiber mats, with representative droplet images (including a PS film shown as a smooth-surface reference) and quantitative contact-angle values reported across electrospinning conditions.
These contact-angle outputs are used to compare hydrophobicity trends with solvent ratio and applied voltage and to parameterize a Cassie–Baxter-based calculation of fractional projected area (f) reported for the fiber mats.
The fiber mats are described as highly hydrophobic based on water contact-angle characterization, with reported contact angles spanning from 130.1° to 143.2° across the studied conditions.
The authors report: “Generally, the contact angle increases as applied voltage increases,” based on the contact angle versus voltage comparison for multiple solvent ratios (Figure 7 and Table 5).
The highest contact angle value in Table 5 is 143.2 ± 0.6° for the chloroform:DMF ratio of 30:70 at 22.5 kV.
The paper explains the voltage-driven contact-angle increase as a combined effect of reduced fiber diameter, increased bead frequency, and pores on the fiber surface, which together increase surface roughness and promote hydrophobic behavior.
Using the Cassie–Baxter model, the authors report fractional projected area (f) values that are less than 0.3 for all samples (Table 6), and highlight sample C:D_30:70_22.5 with f = 0.143 and an “air below the droplet” contribution of 0.857.

Shows contact-angle images of the PS film (smooth surface reference) and electrospun samples made with different chloroform:DMF ratios at 22.5 kV.
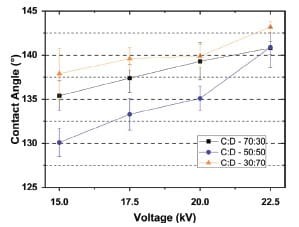
Plots contact angle versus applied voltage for fiber mats produced using the different chloroform:DMF ratios, supporting the stated trend of increasing contact angle with voltage.
Within the paper’s framing, wettability (water contact angle) is used as the quantitative surface metric to connect electrospinning conditions (mixed-solvent ratio and applied voltage) to the hydrophobic performance of porous PS fiber mats.
The authors position these hydrophobic, porous fiber mats as candidates for applications including textiles, filtration, and biomedical fields, with contact-angle results serving as the study’s primary wettability evidence alongside morphology and porosity characterization.
The study uses water contact angle (measured on a Droplet Lab tabletop goniometer) to compare hydrophobicity across mats produced with different chloroform:DMF ratios and voltages.
Contact angle increases with applied voltage across the tested solvent ratios (Figure 7 and Table 5), providing a direct wettability trend linked to the electrospinning parameter sweep.
The highest reported contact angle is 143.2 ± 0.6° for the 30:70 chloroform:DMF condition electrospun at 22.5 kV (Table 5).
The paper reports Cassie–Baxter fractional projected area (f) values (Table 6) and highlights C:D_30:70_22.5 with f = 0.143 and an air contribution under the droplet of 0.857.
The authors attribute contact-angle differences to changes in fiber diameter, bead formation, and pore presence, describing these as contributors to increased roughness and hydrophobic behavior.