Primary surface measurement reported
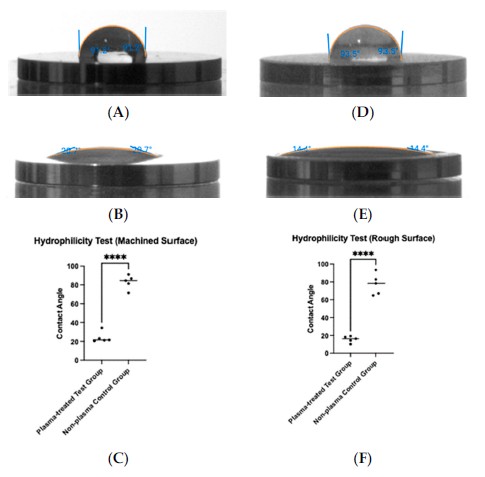
Saline contact angle on titanium discs before and after vacuum plasma treatment, used to evaluate surface hydrophilicity on machined and rough surfaces.
Client Citation Analysis
Saline contact angle on titanium discs before and after vacuum plasma treatment, used to evaluate surface hydrophilicity on machined and rough surfaces.
The wettability test was conducted using “a tensiometer (Droplet Lab, Droplet Biosciences, Cambridge, MA, USA)” to measure the contact angle of a saline droplet on titanium surfaces.
The contact-angle data were used to compare titanium surface hydrophilicity before and after plasma treatment on machined and rough discs. The authors interpreted the lower post-treatment contact angles together with reduced carbon contamination and improved early fibroblast and osteoblast attachment.
Five machined and five rough-surfaced titanium discs were used for wettability testing, with the same discs measured before and after treatment to ensure consistency.
4.2
0.967
0.647
3.9
4.0
The paper reports titanium-surface hydrophilicity by measuring the contact angle of a saline droplet before and after plasma treatment. Reported values decreased from 83.1° to 24.1° on machined discs and from 77.3° to 15.7° on rough discs.
Supporting measurements included SEM surface-topography imaging, EDS-based carbon analysis, cell viability assays for fibroblast and osteoblast adherence, immunohistochemistry staining of actin cytoskeleton and nuclei, SEM imaging of cell morphology, and RNA sequencing for differential gene expression.
The Dropometer appears in the methods as a tensiometer used for a wettability test on titanium discs. The authors placed a saline droplet on the surface and measured contact angle before and after 30 s vacuum plasma treatment on both machined and rough-surfaced Grade 4 titanium discs, using the same discs for paired before/after comparison.
In the study workflow, the Dropometer supplied the direct hydrophilicity readout that the authors used to quantify the plasma-induced surface shift and relate that shift to subsequent cell-response findings.
Plasma treatment produced a strong reduction in saline contact angle on both titanium surface types. Machined discs shifted from 83.1° to 24.1°, and rough discs shifted from 77.3° to 15.7°, with both reductions reported as statistically significant at p < 0.0001.
EDS analysis showed carbon content decreasing from 2.60% to 1.87%, a 28.1% reduction after plasma treatment. SEM imaging at 10,000× showed the titanium surface architecture was preserved during treatment.
Fibroblast adherence on machined titanium discs was higher at the 1, 2, and 6 h time points after plasma treatment. Immunohistochemistry and SEM also showed broader cytoskeletal spread and more spread-out pseudopodia morphology at early time points.
Osteoblast adherence on rough titanium discs was higher at the 1 and 2 h time points after plasma treatment. IHC and SEM images showed more spread-out morphology, and by 24 h osteoblasts in the plasma group displayed a long, spindle-shaped, well-attached morphology.
RNA sequencing identified two genes as significantly upregulated in plasma-treated fibroblasts at 6 h versus the no-plasma group: Apln (log2 fold change = 1.90, FDR = 3.9 × 10−2) and Crabp2 (log2 fold change = 3.13, FDR = 1.4 × 10−4). The paper links these genes to angiogenesis and cell growth differentiation.
This figure shows the saline-droplet contact-angle images and before/after distributions for machined and rough titanium surfaces, making it the central visual for the Dropometer-derived hydrophilicity result.
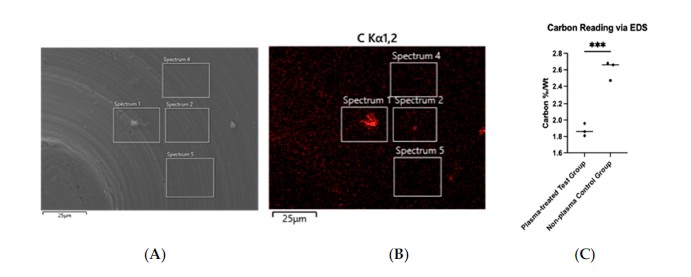
This figure shows EDS-based carbon mapping and the reduction in carbon percentage by weight after plasma treatment, providing chemical context for the hydrophilicity change seen in Figure 5.
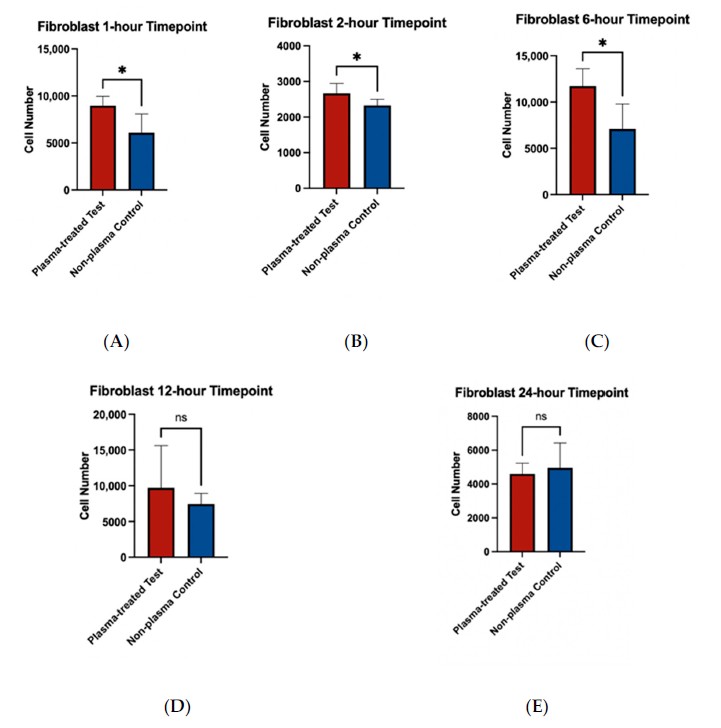
This figure shows that the plasma-treated machined surfaces had higher fibroblast adherence at early time points, connecting the wettability shift to the soft-tissue-facing cell model used in the study.
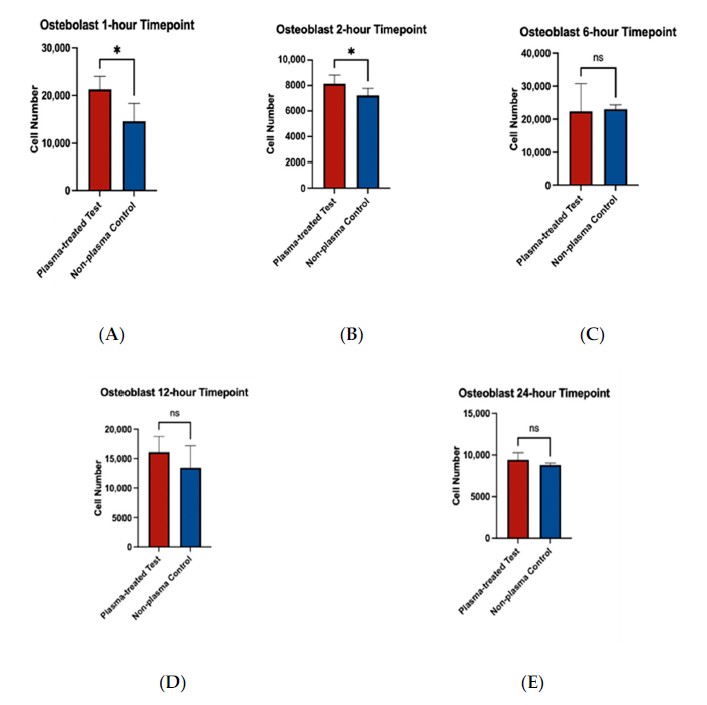
This figure shows higher early osteoblast adherence on plasma-treated rough surfaces, linking the surface hydrophilicity result to the implant-body-facing cell model.
In this paper, the Dropometer-generated contact-angle data are the study’s direct surface-level evidence that vacuum plasma treatment shifts titanium toward a more hydrophilic state. That shift sits at the center of the authors’ interpretation of plasma bioactivation, alongside reduced carbon contamination and preserved surface architecture.
Within the paper’s implant-biomaterials context, the wettability data help connect a fast chairside plasma step with early biological events on titanium, including stronger early fibroblast and osteoblast attachment and a fibroblast gene-expression response at 6 h. The authors frame these results around improved implant biocompatibility and early healing-related interactions on implant and abutment surfaces.
The Dropometer provided the quantitative surface readout that distinguishes untreated from plasma-treated titanium in this study. The contact-angle shift was large on both machined and rough discs.
The same discs were measured before and after treatment, which made the wettability comparison tightly matched to the plasma intervention. That design strengthens the study’s surface-level comparison.
The authors used the Dropometer on both machined and rough titanium surfaces, aligning the wettability test with abutment-like and implant-body-like use cases inside the study design.
In this paper, the contact-angle result gains value when read together with EDS carbon reduction, cell-attachment assays, morphology imaging, and RNA sequencing. The study uses that combined workflow to interpret plasma treatment as a surface-bioactivation step.
The most prominent biological differences appeared at early time points, making the Dropometer readout especially relevant as an upstream indicator of the surface state the cells first encounter.