Primary surface measurement reported
Static water contact angle was measured on control and GO-coated urinary catheter samples by static sessile drop.
Client Citation Analysis
Static water contact angle was measured on control and GO-coated urinary catheter samples by static sessile drop.
The paper states that wettability of coated UC samples was assessed via static sessile drop using “a Droplet Lab system (Brampton, ON, Canada)”.
The contact-angle data were used to compare the control, PEI+GO, and PEI/GO surfaces and to show that the coating processes increased hydrophilicity together with surface roughness. These surface-property results sat alongside FTIR, Raman, microscopy, molecular dynamics, and antibacterial assays in the architecture-dependent comparison.
Wettability was assessed by static sessile drop using 10 µL droplets with n = 5.
5.3
Q3 - Bioengineering
0.735
3.7
3.9
Q2 - Engineering, Biomedical
Static water contact angle was measured for the control, PEI+GO, and PEI/GO catheter samples. Reported values were 98.3 ± 1.0° for the control, 76.4 ± 1.0° for PEI+GO, and 75.5 ± 1.0° for PEI/GO.
The study paired wettability with zeta potential, surface roughness, FTIR, Raman spectroscopy, optical microscopy, VP-SEM, and FEG-SEM to characterize the two coating architectures. Molecular dynamics simulations tracked potential energy and RMSD for GO–peptide and PEI–peptide complexes, and agar diffusion assays against E. coli and E. faecalis were used as a comparative indicator of peptide release from the coatings.
The Dropometer appears in the characterization workflow as the instrument used to assess wettability of coated urinary catheter samples by static sessile drop. The paper specifies 10 µL droplets and n = 5, with the resulting water contact angles reported for the control, PEI+GO, and PEI/GO samples.
In the study’s comparison of coating architectures, those wettability results were used to show that both GO-based coatings shifted the catheter surface toward higher hydrophilicity while the broader dataset separated the layered and embedded structures by morphology, thickness, interaction energetics, and antibacterial response.
The control catheter showed a water contact angle of 98.3 ± 1.0°, while PEI+GO and PEI/GO measured 76.4 ± 1.0° and 75.5 ± 1.0°, respectively. The authors state that both coating processes increased hydrophilicity and roughness.
Microscopy described PEI+GO as porous and cracked, whereas PEI/GO appeared smoother. Cross-sectional SEM gave an average thickness of 320 ± 33 nm for PEI+GO and 890 ± 115 nm for PEI/GO.
Potential-energy profiles stabilized from about 8 ns onward, and the most negative energies corresponded to the PEI complexes with E14LKK and fLFB. The paper interprets this as stronger PEI–peptide stability over time.
The GO–fLFB complex maintained the highest potential energy and showed the greatest RMSD fluctuations among the four modeled complexes. The authors interpret this behavior as consistent with more ready release of fLFB in the GO matrix.
Against both E. coli and E. faecalis, the PEI+GO–fLFB coating produced the largest inhibition zones. The paper links this to weaker GO–fLFB interaction and the layered architecture.
The study’s conclusion is that coating configuration and GO–AMP interaction strength together dictated antimicrobial performance on urinary catheters. In the tested set, PEI+GO outperformed PEI/GO in antibacterial activity.
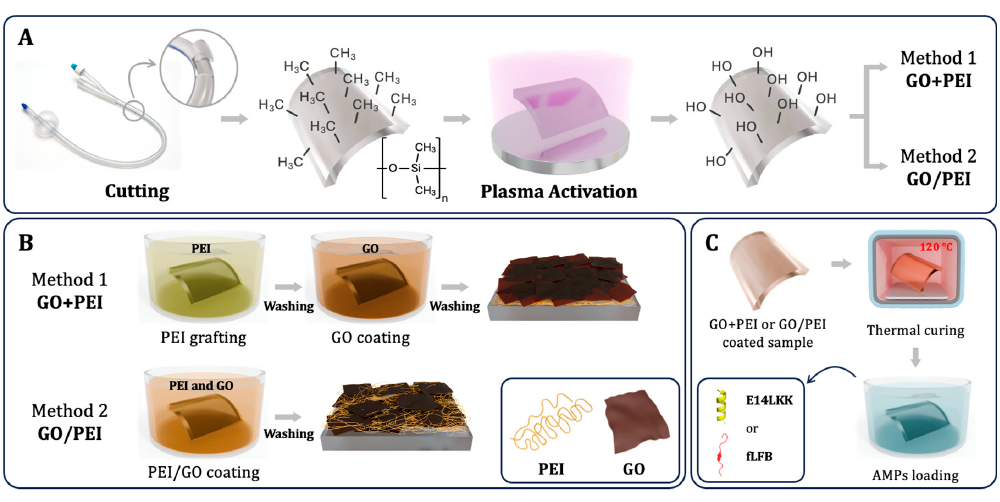
This schematic lays out the plasma pretreatment, the PEI+GO and PEI/GO coating routes, and the later thermal curing and AMP loading steps used in the overall study design.
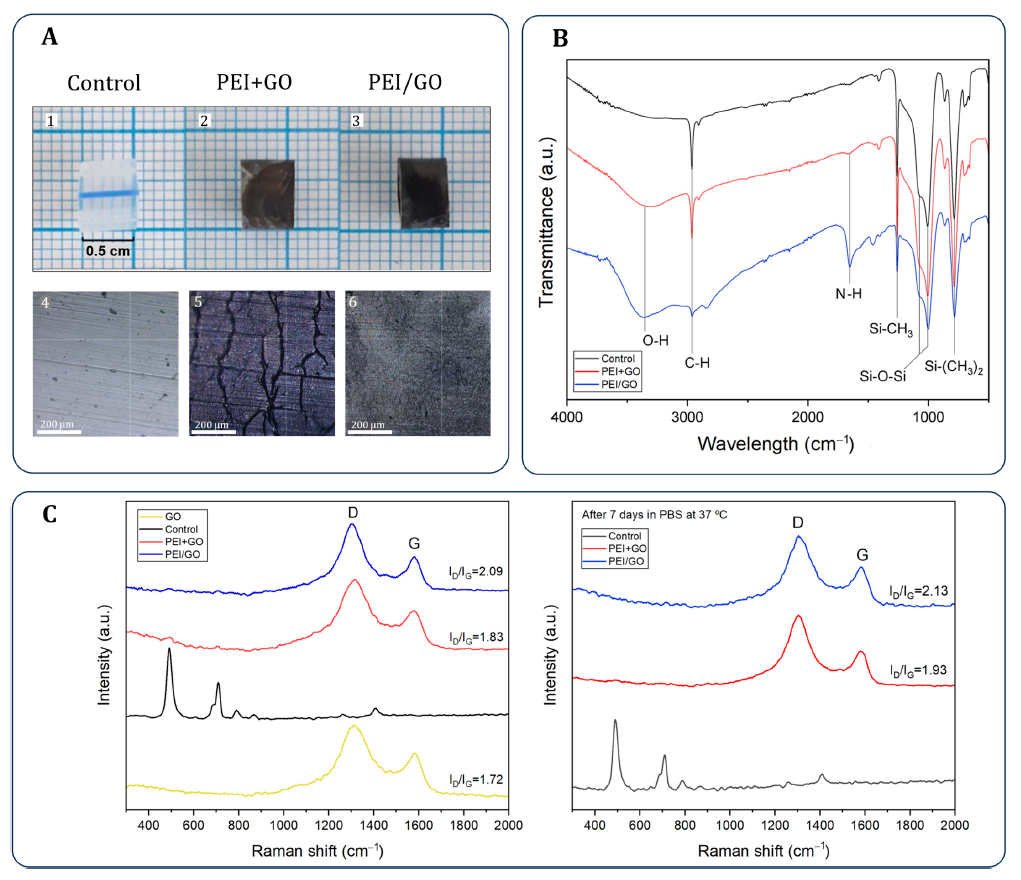
Figure 3B and Table 1 present the wettability and roughness comparison that anchors the Dropometer-derived contact-angle results for control, PEI+GO, and PEI/GO samples.
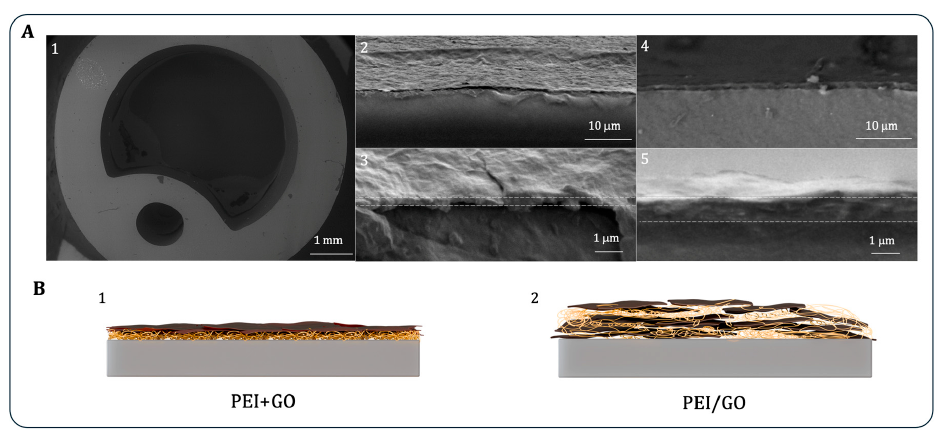
Cross-sectional SEM in Figure 4 shows the layered PEI+GO structure and the embedded PEI/GO structure, giving the physical context for the surface-property differences measured in Figure 3.
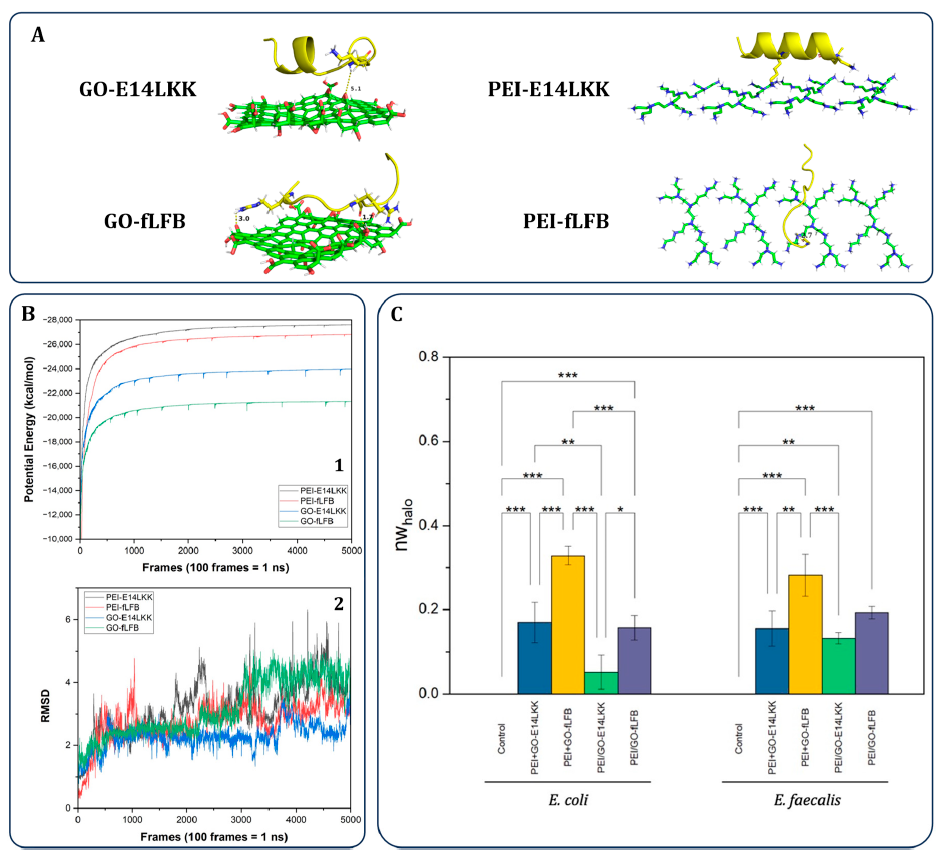
Figure 5 combines molecular dynamics outputs and antibacterial data used by the authors to interpret why the PEI+GO–fLFB system showed the strongest activity.
For this urinary-catheter coating study, the Dropometer-derived contact-angle data establish the surface-state shift that follows GO-based coating deposition. The control surface was markedly more hydrophobic than the two coated surfaces, so wettability became one of the paper’s direct readouts for how the PEI+GO and PEI/GO architectures changed the catheter interface.
Those wettability results matter in the paper because they are read together with roughness, FTIR, Raman, SEM, molecular dynamics, and inhibition-zone data rather than in isolation. That combined view is what the authors use to explain how coating architecture modulates peptide availability and why the layered PEI+GO–fLFB system gave the strongest antibacterial outcome in their comparison.
In this paper, sessile-drop contact angle clearly separated the uncoated catheter from both GO-coated surfaces, making it a straightforward readout of coating-induced surface change.
PEI+GO and PEI/GO had very similar contact angles, yet they differed strongly in morphology and thickness, which became important in the later release and antibacterial interpretation.
The authors did not rely on wettability by itself; they interpreted it alongside roughness, FTIR, Raman, and SEM to characterize the coated surfaces.
The study tied the surface characterization workflow to peptide-interaction simulations and antibacterial assays, using all three layers of evidence to explain architecture-dependent performance.