Primary surface measurement reported
Surface tension of polymer–surfactant solutions measured at room temperature (reported as surface tension vs. surfactant concentration plots).
Client Citation Analysis
Surface tension of polymer–surfactant solutions measured at room temperature (reported as surface tension vs. surfactant concentration plots).
Surface tension was measured using a smartphone-based tensiometer using the ADSA (Axisymmetric Drop Shape Analysis) method
Surface tension vs. surfactant concentration curves (paired with conductivity curves) were used to discuss surface activity and to estimate approximate CAC (critical aggregation concentration) and PSP (polymer saturation point) based on slope changes.
Each solution’s surface tension was measured 12 times and averaged; the authors describe the measurements as “very consistent” and use this to support method reliability.
9.7
1.227
0.918
4.9
5.2
Q1 - Polymer Science (19/94)
Surface tension of aqueous polymer–surfactant solutions at room temperature, obtained from pendant-drop measurements analyzed using ADSA with a smartphone-based tensiometer
Electrical conductivity of polymer–surfactant solutions (used alongside surface tension to interpret concentration-dependent behavior).
Steady-shear rheology (shear stress vs. shear rate and viscosity vs. shear rate), including power-law model parameters K (consistency index) and n (flow behavior index).
The paper uses Dropometer by Droplet Lab a smartphone-based tensiometer to measure surface tension via the ADSA (Axisymmetric Drop Shape Analysis) method. A pendant droplet is formed at the tip of a needle or capillary, back-illuminated, imaged at high resolution using a smartphone camera, and analyzed with specialized software that calculates surface tension numerically from the droplet geometry.
These surface-tension outputs are used throughout the “surface activity” results to compare surfactant effectiveness and to estimate approximate CAC and PSP values from changes in slope of surface-tension–concentration plots (often considered alongside conductivity plots).
Across polymer–surfactant mixtures, the surface tension decreases as surfactant concentration increases and tends to level off at higher concentrations. The authors interpret the initial decrease as adsorption at the air–water interface and the leveling behavior as saturation of the interface by surfactant–polymer complexes and surfactant molecules.
In the cross-surfactant comparisons (LR-400: Figure 19b; Praestol 2540TR: Figure 25b), Amphosol is identified as the most effective surfactant in reducing surface tension at a given concentration.
For surfactant/LR-400 polymer solutions, at any given surfactant concentration, the surface tension order reported is: Stepwet > Stepanol > HTAB > Alfonic > Amphosol.
For surfactant/Praestol 2540TR polymer solutions, at any given surfactant concentration, the surface tension order reported is: HTAB > Stepanol ≥ Stepwet > Alfonic > Amphosol.
The authors use changes in slope of surface tension vs. concentration plots (and paired conductivity plots) to estimate CAC and PSP values, and note that the plots do not show “sharp enough breaks” to draw definite conclusions; they also mention alternative methods (e.g., calorimetry or spectroscopy) as possible validation approaches.
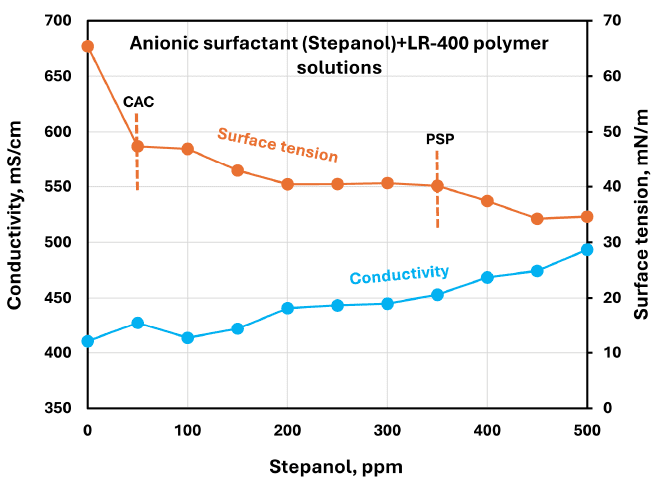
Shows electrical conductivity and surface tension versus Stepanol concentration in surfactant + LR-400 solutions, with CAC/PSP indicated in the plotted trends.
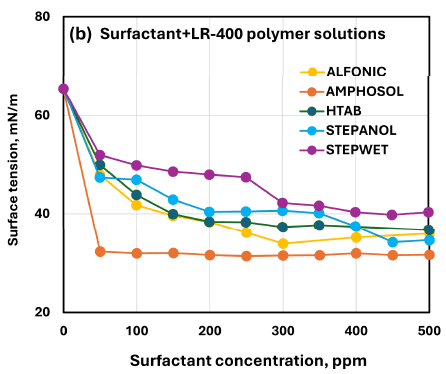
Compares surface tension behavior across the five surfactants at matched concentration in the LR-400 polymer solution, including the reported surface-tension ordering and identification of Amphosol as most effective.
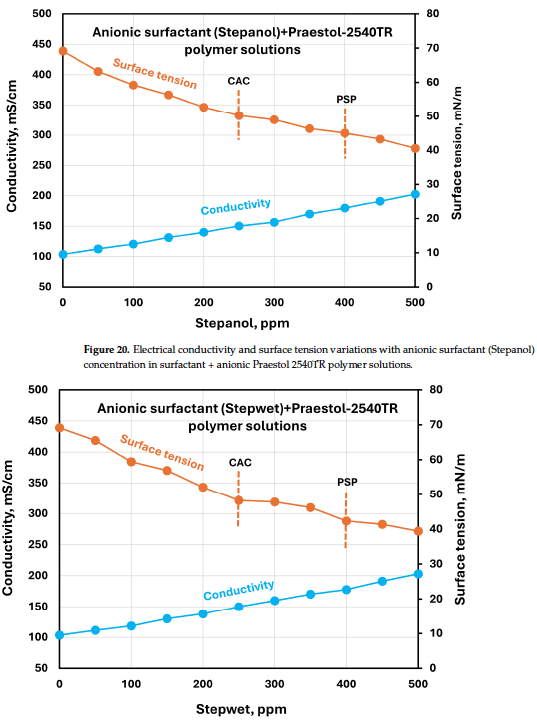
Shows electrical conductivity and surface tension versus Stepanol concentration in surfactant + Praestol 2540TR solutions, illustrating the concentration dependence used for CAC/PSP estimation.
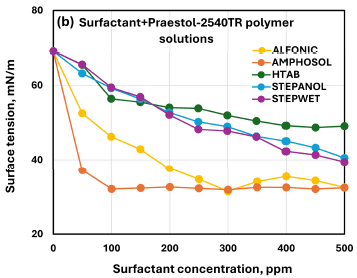
Compares surface tension behavior across the five surfactants at matched concentration in the Praestol 2540TR polymer solution, including the reported surface-tension ordering and identification of Amphosol as most effective.
The paper frames polymer–surfactant interactions as important for designing advanced fluid systems used in applications such as enhanced oil recovery, drilling, and chemical processing. Within that scope, the surface-tension measurements provide the “surface activity” component of the study, enabling direct comparison of how surfactant type influences the air–water interface behavior across the two polymer systems.
By pairing surface tension with conductivity versus surfactant concentration, the authors use the Dropometer (smartphone-based tensiometer with ADSA) to interpret adsorption/saturation behavior and to estimate approximate CAC/PSP regime markers for the tested polymer–surfactant combinations.
Amphosol CG is identified as the most effective surfactant for reducing surface tension at a given concentration in both LR-400 and Praestol 2540TR polymer solutions.
The paper describes surface tension curves that drop sharply at low surfactant concentration and level off at higher concentration, interpreted as interface saturation behavior.
The authors state the plots do not exhibit sharp enough breaks for definite CAC/PSP determination and present CAC/PSP values as approximate, with calorimetry/spectroscopy mentioned as potential validation approaches.
The paper notes that for Amphosol and Alfonic the surface tension drops sharply then levels off, while for Stepanol, Stepwet, and HTAB the decrease is more gradual in the Praestol 2540TR system.