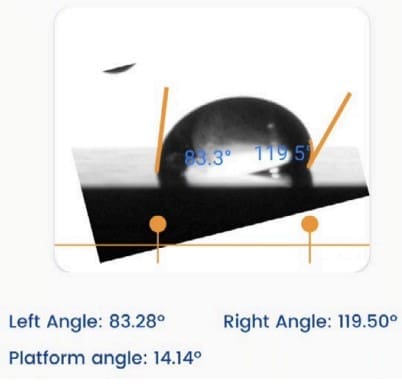
A Droplet Lab Dropometer was used to measure water contact angle to confirm that the hydrophobicity of the Parafilm® layer remained essentially unchanged after the laser ablation step (i.e., the fabrication process did not degrade the barrier’s wettability performance). Specifically:
- In Methods (Device characterization), the paper states that water contact angle of pristine Parafilm® and the Parafilm® layer after laser ablation of paper was assessed using Dropometer (Droplet Lab).
In Results, they report contact angles of ~108° (pristine) vs ~109° (post-ablation), supporting that the PHLC process preserved Parafilm® hydrophobic behavior critical for reliable microfluidic containment.