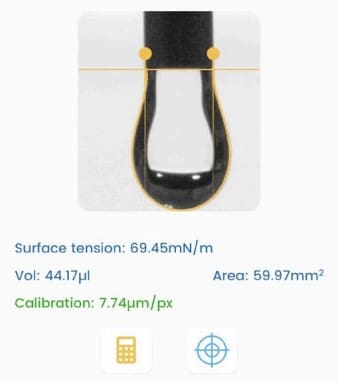
Droplet Lab is used for pendant-drop tensiometry to quantify mineral oil / aqueous PBS interfacial tension for different lipid-containing oil formulations. These values are then interpreted against vesicle formation yield trends, making the Droplet Lab measurement a formulation-screening metric rather than a purely descriptive material property.
What this enables for pharma teams:
- Objective comparison of lipid formulation “interfacial activity” (how strongly a lipid reduces oil/water interfacial tension)
A quantitative bridge from physicochemical measurement → process outcome (yield), supporting earlier-stage down-selection and comparability thinking.