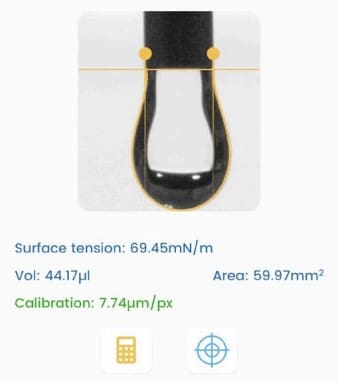
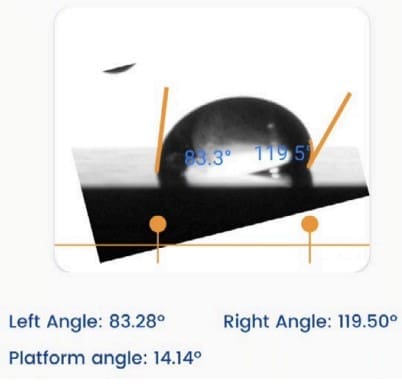
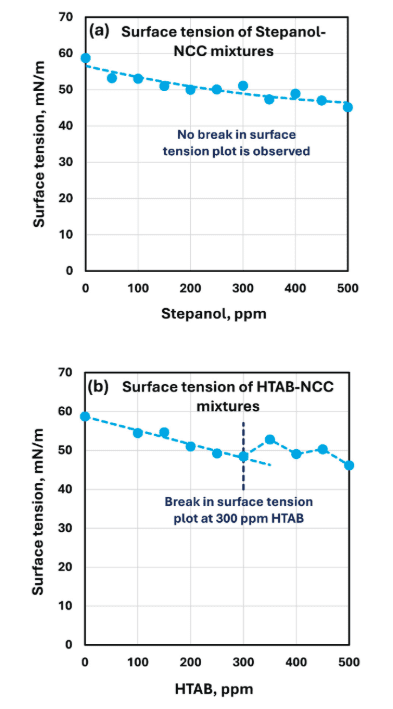
In this work, a Droplet Lab smartphone-based pendant drop tensiometer was used to measure surface tension of:
- NCC dispersion (1 wt%) and
- surfactant–NCC mixtures across 0–500 ppm surfactant.
Paint-relevance of this measurement:
Surface tension is a formulation-critical lever for wetting, flow/leveling, defect control (craters/fisheyes), pigment dispersion behavior, and foaming tendency in waterborne paints. Here, surface tension trends were used as a practical indicator of surfactant adsorption/migration behavior that coincided with major rheology shifts, exactly the type of compatibility signal paint formulators want early in screening.