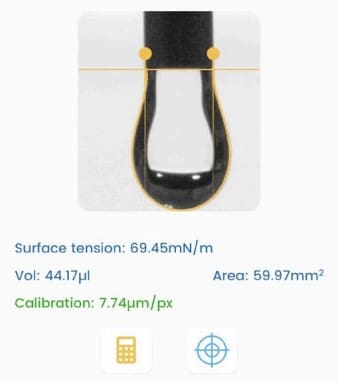
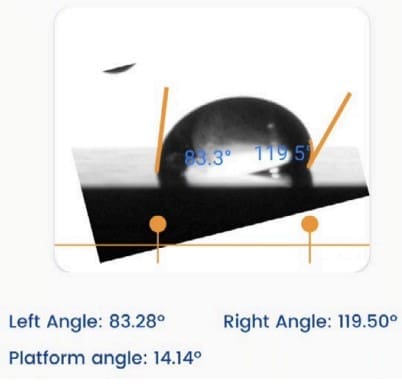
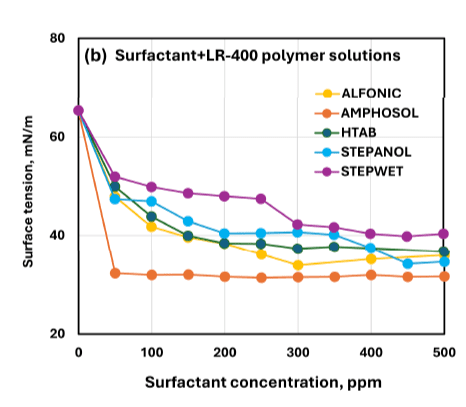
- The Droplet Lab instrument was used specifically for surface tension measurement (not contact angle) via a smartphone-based tensiometer using ADSA (Axisymmetric Drop Shape Analysis) on pendant droplets. The Droplet Lab measurement enables quantifying how surfactant type and concentration shift interfacial properties of polymer–surfactant fluids—critical for designing oilfield fluids where interfacial behavior influences wetting/cleanup and formulation performance.
- Where it’s mentioned in the paper: Surface tension method and Droplet Lab instrument are described in Section 2.4 “Surface Tension Measurements” (page 5), including the ADSA pendant-drop workflow and repeat measurements.