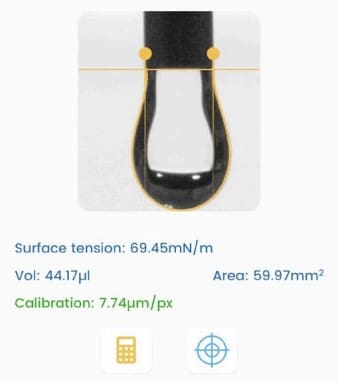
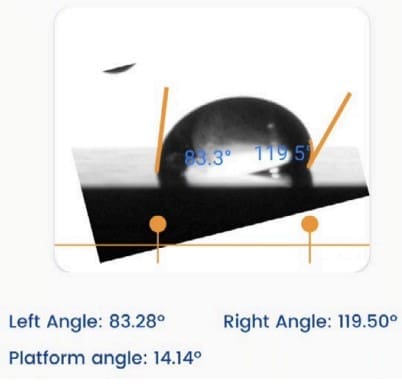
The Droplet Lab goniometer (cited in the paper as “Droplet Smart Tech, Markham, ON, Canada”) was used to measure static contact angles on polyserotonin-coated glass films using 20 µL droplets of milli-Q water, glycerol, and diiodomethane (Materials & Methods, Section 2.2, page 3). Those contact angle measurements were then used to calculate surface energy via the Owens–Wendt–Rabel–Kaelbel (OWRK) model, enabling quantitative comparison of buffer-dependent film wettability and surface energetics (Results/Discussion, Section 3.4, pages 7–8; Supplementary Figure S5 and Table S1 are referenced for the contact angle and surface energy data).